Visible Light-
|
All electromagnetic radiation is light, but we, humans, can only see a small portion of this radiation—this portion is what is called visible light or white light.
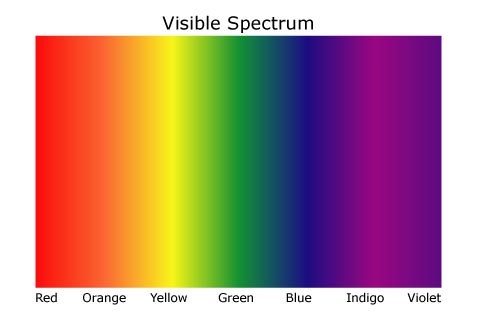
Cone-shaped cells in our eyes act as receivers tuned to the wavelengths in this narrow band of the spectrum. Other portions of the spectrum have wavelengths too large or too small and energetic for the biological capacity of our perception. As the full spectrum of visible light or white light travels through a prism, the wavelengths separate into the colour of the rainbow because each colour is a different wavelength, this is known as dispersion. Violet has the shortest wavelength, at around 400 nanometers and is bent the most, whilst red has the longest wavelength, at around 700 nanometers and is bent the least. Light comes in a range of wavelengths that we can see. Each wavelength is identified as a different colour. These colours are:
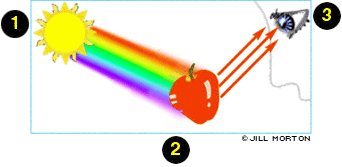
Fact: Blu-Ray Discs The benefit of using a blue-violet laser (405nm) is that it has a shorter wavelength than a red laser (650nm), which makes it possible to focus the laser spot with even greater precision. This allows data to be packed more tightly and stored in less space, so it's possible to fit more data on the disc even though it's the same size as a CD/DVD. From Blu-ray Website; view Bibliography. Visible light is an essential part of life on Earth. Plants absorb this light and use the energy to make carbohydrates, fats, proteins, vitamins and other materials that both humans and animals are dependant on. Seeing in Colour (see Fig. 1, right) 1. All the invisible light or white light rays of the sun shine on to (eg) a red apple; 2. The surface of the apple abosrobs all of the light rays, except those that are related to the colour red; 3. Which is reflected back to our eyes and sent to our brain, giving the apple its red colour. Polarisation and Interference of Light Light travels in an electromagnetic wave in three dimensions. If you wear a polarised pair of glasses on a sunny day, you're still able to see but the lenses of the glasses absorb much of the light energy that hits them. This means that the light has been polarised. Light that is vibrating in a certain direction pass through the filter, and those vibrating in other directions are absorbed. |
|
In today's society we use:
Photovoltaic Cells (solar power)
The photovoltaic cells now used to turn sunlight into electricity can only absorb and use a small fraction of that light, and that means a significant amount of solar energy becomes lost.
Sunlight is made up of many wavelengths of light. In a traditional solar panel, silicon atoms are hit by sunlight and the atoms outermost electrons absorb energy from some of these wavelengths of sunlight, causing the electrons to become excited.
Once the excited electrons absorb enough energy to jump free from the silicon atoms, they can flow independently through the material to produce electricity.
This is called the photovoltaic effect—a process that takes place in a solar panel's photovoltaic cells.
Today, Harry Atwater and his colleagues of Caltech, have found a way to absorb and make use of the infared waves as well as visible waves, with a panel made not of silicon, but rather entirely of metal.
Although silicon-based photovoltaic cells can absorb light wavelengths that fall in the visible spectrum, longer wavelengths such as infrared light pass through the silicon. These wavelengths of light pass right through the silicon and never get converted to electricity—and in the case of infrared, they are normally lost as unwanted heat.
Photovoltaic Cells (solar power)
The photovoltaic cells now used to turn sunlight into electricity can only absorb and use a small fraction of that light, and that means a significant amount of solar energy becomes lost.
Sunlight is made up of many wavelengths of light. In a traditional solar panel, silicon atoms are hit by sunlight and the atoms outermost electrons absorb energy from some of these wavelengths of sunlight, causing the electrons to become excited.
Once the excited electrons absorb enough energy to jump free from the silicon atoms, they can flow independently through the material to produce electricity.
This is called the photovoltaic effect—a process that takes place in a solar panel's photovoltaic cells.
Today, Harry Atwater and his colleagues of Caltech, have found a way to absorb and make use of the infared waves as well as visible waves, with a panel made not of silicon, but rather entirely of metal.
Although silicon-based photovoltaic cells can absorb light wavelengths that fall in the visible spectrum, longer wavelengths such as infrared light pass through the silicon. These wavelengths of light pass right through the silicon and never get converted to electricity—and in the case of infrared, they are normally lost as unwanted heat.